HYPERGLOBULINEMA AND RISK ASSESSMENT: A CASE STUDY

Sheetal Salgaonkar, MD VP & Medical Director, Global Medical RGA India ssalgaonkar@rgare.com
[Reprinted with permission from RGA]
Serum Proteins
Total serum protein is a test commonly administered as part of the individual insurance application process. It measures the total amount of protein in the serum, as well as providing amounts of serum albumin and globulin, the two major types of blood proteins. The typical reference range for this test is 6 to 8 g/dl.
Albumin, which is made in the liver, is the most abundant protein in serum, with a reference range of 3.5 to 5 g/dl. Globulin generally comprises a much smaller fraction, with a reference range of 1.5 to 3.0 g/dl, and as total globulin values are generally arrived at by subtracting the albumin number from the total protein value, the certainty of globulin measurement is limited by the accuracy of the method used to calculate total protein and albumin.
Globulin contains many important proteins, such as carrier proteins, enzymes, complement and immunoglobulins. Most of these are synthesized in the liver, while immunoglobulins are synthesized by the plasma cells.
Most instances of elevated serum protein stem either from dehydration, in which blood volume decreases, thereby concentrating proteins, and increased production of specific proteins, which is more common. Immunoglobulins are the most commonly overproduced proteins and are generally elevated in the presence of infection and hematological neoplasm.1
Elevated globulin levels require underwriter awareness, as they may indicate disease states, and thus higher mortality risk. A 2014 study by Fulks et al., for example, found that persons with globulin values greater than 3.2 g/dl experience excess mortality, and those with values of greater than 4.0 g/dl have approximately double the excess mortality risk across all ages and sexes.22Protein levels both higher or lower than the reference range can also indicate other causes, including pregnancy, diabetes, anemia and more.
Executive Summary
Total serum protein, along with its components serum albumin and globulin, are frequently encountered in insurance applications. The globulins, though the smaller fraction, can be a red flag for underwriters, for when elevated they can indicate impairments such as infections, chronic inflammation, or neoplasia. An elevated globulin level is generally a clinical indication to order a serum protein electrophoresis (SPEP). The main clinical value of an SPEP is to determine which of the four globulin proteins is elevated, and to differentiate between monoclonal and polyclonal gammopathies. The differentiation is vitally important, as monoclonal gammopathies, which are indicated by evidence of a monoclonal (M) band on the SPEP, may indicate malignant or premalignant conditions. This article presents an interesting case study of an elevated serum globulin incidentally detected in the course of an insurance application process. The cause turned out to be monoclonal gammopathy of unknown significance (MGUS), which is the most common monoclonal gammopathy.
The albumin/globulin (A/G) ratio is an additional metric that can be used to indicate disease states. It is not a specific marker, however, as it can change if albumin or globulin levels increase or decrease. It is important to be aware that an A/G ratio of less than 1 is clinically significant, as it can indicate either that globulin production has increased to a level exceeding that of albumin, or that albumin production has reduced or ceased.
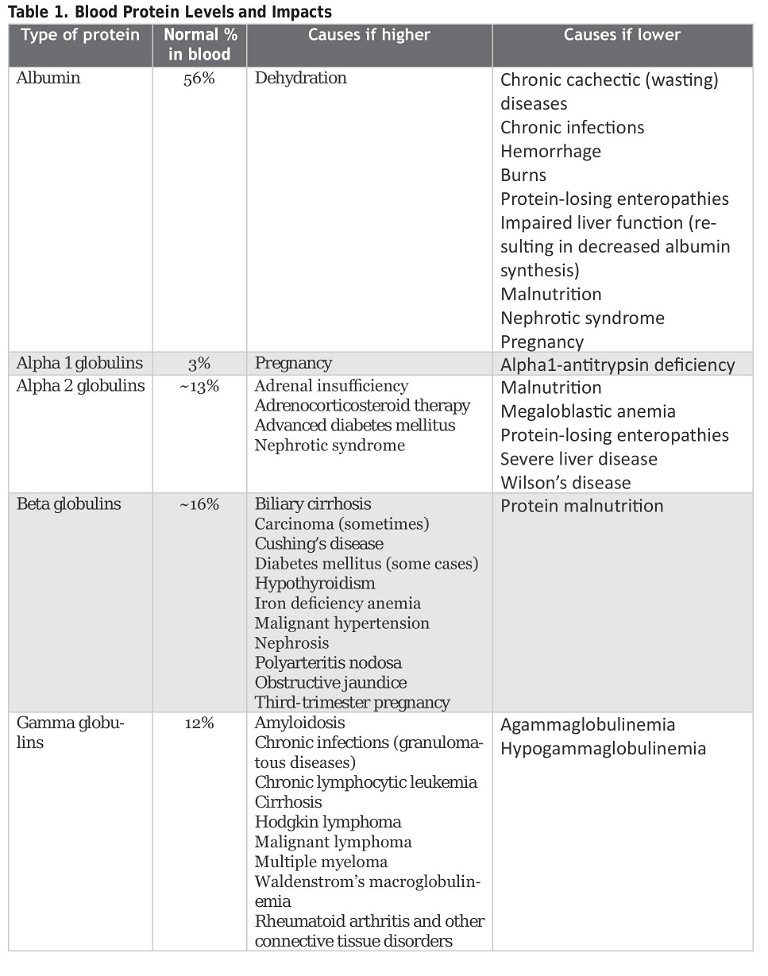
Table 1 (below) lists the many impairments related to blood protein level abnormalities.3
At this point, let us consider the applicant:

He underwent a series of blood tests as part of his life insurance application process, which yielded the following results:
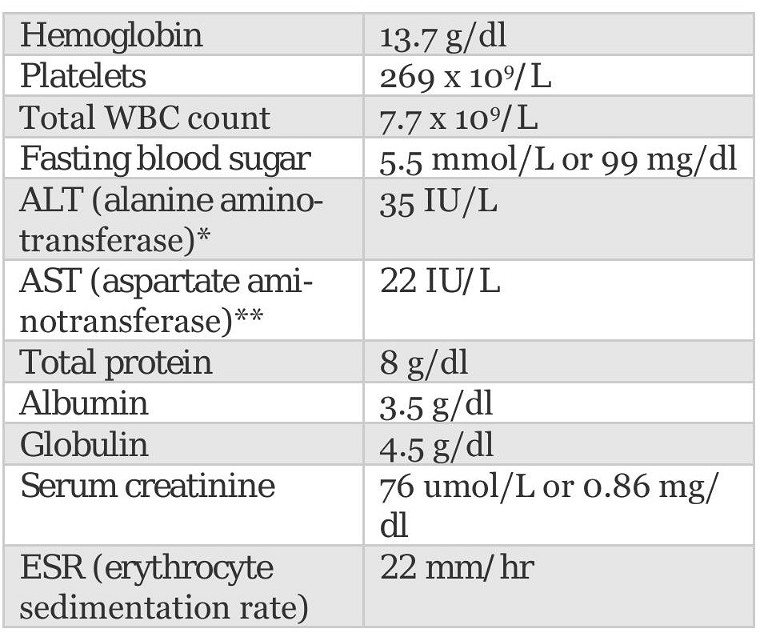
*Can also appear as SGPT (serum glutamic pyruvic transaminase)
**Can also appear as SGOT (serum glutamic oxaloacetic transaminase)
Primary abnormality: Although the total serum protein result of 8 g/dl is still within normal range, the globulin result of 4.5 g/dl, which is higher than the reference range, is concerning.
Differential diagnosis: Elevated globulins can indicate infections, chronic inflammation or neoplasia.
Which further investigation(s) would be helpful? In general, an elevated total serum protein level – something usually found incidentally – is a clinical indication to order serum protein electrophoresis (SPEP). In this particular case, even though total serum protein was within the normal range, the elevated globulin would prompt a request for further testing.
About Serum Protein Electrophoresis
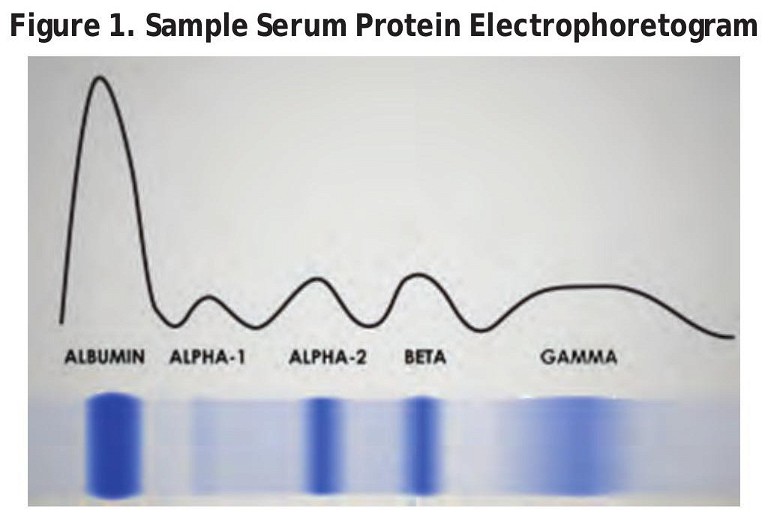
SPEP is specifically used to separate and analyze amounts of the five main blood serum proteins: albumin, and the four types of globulins – alpha 1 globulin, alpha 2 globulin, beta globulin and gamma globulin. The test separates the proteins based on physical properties such as molecule size and strength of electrical charge.
To conduct the test, serum is placed upon a specific medium and an electrical charge is then applied. Albumin molecules, which are larger and have a strong net negative charge, will migrate along the x axis toward the positive electrode. Globulins, being smaller molecules with weak negative charges, will move toward the negative electrode with the gamma peak closest to the negative electrode. The amounts of each protein are then measured.
The bands and graph seen in the SPEP in Figure 1 (below) depicts the amounts of albumin and the four main globulins found in a healthy individual.
As for the applicant, his elevated serum globulin level caused his case to be postponed for further evaluation. He consulted a hematologist, who advised him to undergo additional blood tests, including an SPEP, which together determined a diagnosis of monoclonal gammopathy of unknown significance (MGUS). The applicant then submitted this additional information to the insurance company for further review.
The results of the applicant’s follow-up blood tests are in Table 2, below.

Key questions to be addressed during underwriting:
• What does this diagnosis mean?
• What is the applicant’s risk of progression to multiple myeloma?
• Can life insurance be offered?
Gammopathy Types
A gammopathy is defined as a higher than normal level of gamma globulin in the blood. Gammopathies can be either polyclonal or monoclonal. Distinguishing between the two types is of paramount importance when assessing both health and insurability.
• Polyclonal gammopathies: These are characterized by increased levels of more than one type of immunoglobulin, and usually signify nonmalignant conditions such as inflammatory or reactive disorders.3 This diagnosis is associated with a broad or diffuse peak in the gamma globulin band of the SPEP (as seen in Figure 1).
The most common causes of polyclonal gammopathies are infections. These can include: parasitic infections such as malaria, schistosomiasis, strongyloidiasis and leishmaniasis; fungal infections such as paracoccidiodomycosis and histoplasmosis; bacterial infections such as brucellosis and rickettsial conditions; and spirochete infections such as borrelia (Lyme disease).
They can also be present in certain viral infections such as hepatitis, varicella zoster (chicken pox) and Epstein-Barr, as well as in chronic liver disease, autoimmune diseases such as rheumatoid arthritis and Sjogren’s syndrome, acquired immune deficiency syndrome (AIDS), nonhematologic cancers, and certain malignant lymphomas and lymphoproliferative disorders.4
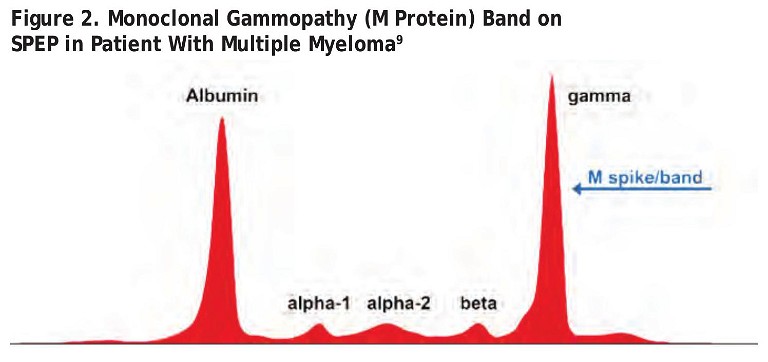
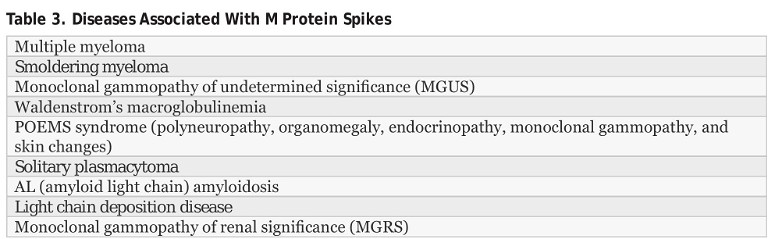
• Monoclonal gammopathies: The critical hallmark of a monoclonal gammopathy is the presence on an SPEP of a sizable and narrow spike in the gamma globulin band. That spike, called an M (for monoclonal) band (or spike) or M protein or paraprotein, is caused by the clonal proliferation of plasma cells. It is associated with malignant or premalignant conditions, as listed in Table 3 (next page). 5
If a monoclonal spike is present in an SPEP’s gamma region, laboratories will usually perform immunofixation, a method to determine the spike’s specific immunoglobulin class (most often IgG or IgA; rarely IgM, D, or E). It is also important to then determine the types and amounts of free light-chains and the serum free light-chain assay (FLC) ratio, in order to diagnose the possible impairment.
Human immunoglobulin has two identical light chains – kappa and lambda – each of which is encoded on a different chromosome. In healthy individuals, the majority of serum light chains bind to heavy chains to form antibodies, so normal serum will have low free light-chain and heavy-chain amounts.
The ratio (or proportion) of kappa to lambda lightchains in serum indicates whether there is an excess amount of one type of light-chain vs. the other, and so can be used as an indicator of diseases specific to each as well as disease progression or remission.
A kappa-lambda ratio of between 0.26 and 1.65 is considered normal. A ratio of less than 0.26 indicates overproduction of lambda light-chains, whereas one higher than 1.65 indicates overproduction of kappa light-chains.
About MGUS
MGUS is the most common monoclonal gammopathy. It is a clinically asymptomatic premalignant clonal plasma cell or lymphoplasmacytic proliferative disorder. MGUS is often discovered as an incidental finding while undergoing routine blood tests or evaluation for other conditions. Its prevalence increases with age, affecting up to 2% of persons age 50 or older and about 3% of those older than age 70.6
The International Myeloma Working Group’s criteria for diagnosing MGUS are as follows:7
• <30 g/L serum paraprotein (monoclonal M protein).
• <10% clonal bone marrow plasma cells.
• No myeloma-defining events (e.g., hypercalcemia, renal insufficiency, anemia, or bone lesions).
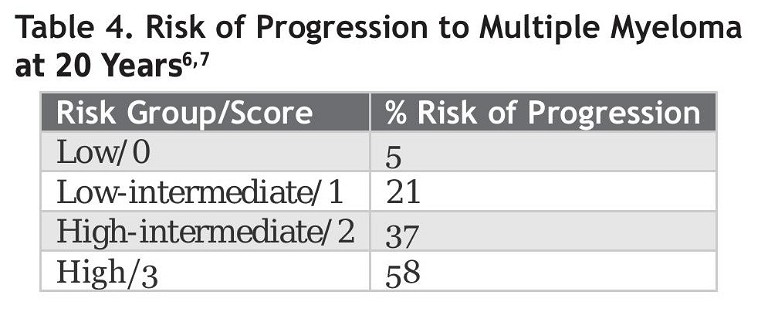
A diagnosis of MGUS is important, as patients can be at risk of progressing to multiple myeloma. The average risk of progression is 1% per year.6,76,7The following three factors can be considered adverse risks in MGUS. In the criteria, each factor is given a score of 1 for calculating the risk group.
• Type of paraprotein: IgA and IgM (both are associated with a higher risk of progression).
• Mprotein concentration: >1.5 g/dl.
• Abnormal serum FLC (kappa-lambda) ratio: <0.26 or >1.65.
Individuals diagnosed with MGUS therefore need to be followed up regularly. Current guidelines recommend follow-up blood tests every 3 to 4 months in the first year, followed by monitoring every 6 to 12 months thereafter.8
Case Assessment
In the case presentation, given that the diagnosis was of an IgG MGUS, and the paraprotein level was 0.83g/dl and serum FLC ratio was 0.7, he is in the low-risk group with a score of 0, indicating a 5% risk of progression at 20 years. Therefore, we can offer life insurance at favorable rates.
Conclusions/Key Takeaways
• Elevated globulin levels can indicate infections, chronic inflammation or hematological neoplasm.
• Polyclonal gammopathies usually indicate nonmalignant conditions, whereas monoclonal gammopathies indicate premalignant or malignant conditions. Hence, distinguishing them is of paramount importance.
• The critical hallmark of a monoclonal gammopathy is evidence of a monoclonal (M protein or paraprotein) spike in the gamma globulin band of an SPEP.
• MGUS is the most common monoclonal gammopathy and is often discovered as an incidental finding on routine screening tests.
• Risk stratification of MGUS (i.e., whether it will progress to multiple myeloma and if so, how quickly) can be based on the type and concentration of monoclonal paraprotein detected as well as the serum FLC (kappa-lambda) ratio.
Notes
1. Bertholf RL. Proteins and Albumin. LabMedicine (Oxford Academic). 2014 Feb 1; 45(1): e25-e41. https://academic.oup.com/labmed/article/45/1/e25/2657886?login=true.
2. Fulks M, Stout RL, Dolan VF. Serum Globulin Predicts All-Cause Mortality for Life Insurance Applicants. J Insur Med. 2014; 44(2): 938. www.crlcorp.com/wp-content/uploads/2016/11/Serum-Globulin-Predicts-All-Cause-Mortality-JIM.pdf.
3. Ramanathan S, Srinivas CN. Serum Protein Electrophoresis and Its Clinical Applications. Biochemical Testing – Clinical Correlation and Analysis. 2019 Oct 11. www.intechopen.com/chapters/69537.
4. Greipp PR. Hypergammaglobulinemia. In Encyclopedia of Immunology (Second Edition), 1998. www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/hypergammaglobulinemia.
5. O’Connell TX, Horita TJ, Kasravi B. Understanding and Interpreting Serum Protein Electrophoresis. Am Fam Physician. 2005 Jan 1; 71(1): 105-12. https://pubmed.ncbi.nlm.nih.gov/15663032/.
6. Kyle RA, et al. A Long-Term Study of Prognosis in Monoclonal Gammopathy of Undetermined Significance. N Engl J Med. 2002 Feb 21; 346(8): 564-9. www.nejm.org/doi/full/10.1056/nejmoa01133202.
7. Rajkumar VS. MGUS and Smoldering Multiple Myeloma: Update on Pathogenesis, Natural History and Management. Hematology Am Soc Hematol Educ Program. 2005; 340-5. https://pubmed.ncbi.nlm.nih.gov/16304401/.
8. Bird J, et al. UK Myeloma Forum (UKMF) and Nordic Myeloma Study Group (NMSG): Guidelines for the Investigation of Newly Detected M-Proteins and the Management of Monoclonal Gammopathy of Undetermined Significance (MGUS). British Journal of Haematology. 2009 Sep 14; 147(1): 22-42. https://onlinelibrary.wiley.com/doi/full/10.1111/j.1365-2141.2009.07807.x.
About the Author
Sheetal Salgaonkar, MD, FALU, MBBS, DBIM is Vice President and Medical Director with RGA and a member of RGA’s Global Medical team. Based in Mumbai, India, she provides underwriting and claims consultation for RGA’s regional offices in the International division, and is involved in product development, medical underwriting training and development of guidelines for RGA’s Global Underwriting Manual. Dr. Salgaonkar has been a part of the Federation of Indian Chambers of Commerce & Industry’s Task Force for Critical Illness and was a member of the subcommittee for Policy Formulation on Financial Inclusion of Persons with Disabilities for India’s insurance industry. She is also Treasurer of the Indian Insurance Medical Officer’s Association (IMOK) and was the scientific chair of the International Committee for Insurance Medicine’s 2019 meeting, which was held in Mumbai. She has made significant contributions to the course curriculum of the Underwriting Diploma and the Advanced Underwriting Diploma, a joint initiative of the Association of Insurance Underwriters and the Insurance Institute of India.
SCENES FROM THE 2023 AHOU CONFERENCE IN HOLLYWOOD, FL

